Issue 47 – Spring 2020
Issue 47 - Spring 2020
Bringing The Latest In Paediatric Endocrinology To You
2344 Likes
Editorial
Welcome
Thanks to our excellent speakers, we are fortunate in this issue of ESPE News to have
insights into several of the talks you will be able to enjoy at ESPE 2021 Online. On page 6, Ali Abbara and Waljit Dhillo review our
understanding of kisspeptin’s role in puberty, with exciting potential developments in its therapeutic and diagnostic use. Meanwhile, on page 7, Peter Kühnen examines the melanocortin
4 receptor (MC4R) agonist setmelanotide as a treatment option in rare obesity syndromes. He explains the capacity of MC4R agonists to activate
different downstream signalling cascades (‘biased signalling’) and therefore elicit a range of effects. Supporting transgender/gender diverse youth
remains a complex and topical area of healthcare. Stephen Rosenthal discusses the associated issues, which he will address in his forthcoming presentation (page 8).
You can find out more about ESPE 2021
Online at www.eurospe.org/espe2021online.
As always, your contributions will form a central part of the meeting, so please make sure to submit your abstracts by 10 May 2021.
On page 4, we are extremely pleased to have contributions from colleagues in India about their lives in the time of COVID. Researchers
Anuradha Khadilkar and Vandana Jain reflect on the pandemic’s impact on their work with patients and other aspects of their research and daily
lives.The rest of the issue is bursting with the opportunities and support available to you from
ESPE. These extend from grants and committee vacancies to the prospect of future events such as ESPE Schools and the postponed ESPE Science
Symposium. Read on to learn more!We thank all this issue’s contributors for writing for us at such a busy and stressful time.
We wish them, you, and all your families and friends, health and peace in the coming months.
Sarah Ehtisham
Editor, ESPE News
[email protected]
News
Intro
Follow ESPE
online…
Keep an eye on
the latest ESPE news
and activities at
www.eurospe.org
You can also follow
ESPE on Facebook
and Twitter/EuroSPE
News
Opportunities
Council vacancies Be at the heart of ESPE’s strategic decision making and help shape the field of paediatric endocrinology through its activities. Apply now if you are interested in joining ESPE’s Council in September, as: • Science Committee Chair • Communication Committee Chair or • Strategic & Finance Committee Chair. Apply by 2 April 2020 www.eurospe.org/about/vacancies/ council-vacancies-2020 Committee vacancies NEWSLETTER EDITORIAL BOARD Could you be part of the friendly, collaborative and productive team that creates and collates content for this popular newsletter? We would love to hear from you if you are interested in sourcing features on fascinating, cutting-edge research. Board members also help to write short hot topics (around three per year), so we’re looking for someone comfortable with medical writing. The team receives lots of support from the ESPE Team and the Newsletter Project Manager. Apply by 30 April 2020 www.eurospe.org/about/vacancies OTHER VACANCIES We are also currently welcoming applications for members of our Communication, Clinical Practice and Andrea Prader Committees. All ESPE vacancies are advertised at the web address above.
News
Grants & Awards
NEW! Undergraduate Achievement Award This award aims to attract undergraduates towards a career in paediatric endocrinology. Please encourage and sponsor applications from students with a research interest in the field. • €750 grant plus free registration for ESPE 2020 • 6 grants available each year • requires submission of an abstract to ESPE 2020 and a supporting statement from an ESPE member Apply by 15 April 2020 www.eurospe.org/grants-awards/grants/ undergraduate-achievement-award Research Fellowship Open to ESPE members based in Europe who can demonstrate an interest in, and potential for, performing high quality research in paediatric endocrinology. One successful applicant will be awarded a grant of up to €140,000 to conduct research at a leading institution. Watch videos of two previous winners describing the impact of the Fellowship on their careers at the web address below. Apply by 15 April 2020 www.eurospe.org/grants-awards/grants/ research-fellowship Clinical Fellowship Open to those wishing to pursue a career in paediatric endocrinology who have limited training opportunities in their home country. Successful applicants will be supported to undertake practical, clinical training at a European centre of excellence. Apply by 31 May 2020 www.eurospe.org/ grants-awards/grants/ clinical-fellowship
News
Events
We thank Malcolm Donaldson (UK), co-founder of the ESPE Maghreb School, who is stepping down as Steering Committee Co-ordinator. Malcolm is pictured with Asmahane Ladjouze (Algeria), the new Co-ordinator for 2020. EVENTS Asmahane Ladjouze and Malcolm Donaldson John Gregory SEE PAGES 8−10 FOR MORE EVENTS NEWS: > ESPE 2020 Liverpool, UK 10−12 September 2020 > ESPE Science Symposium Nijmegen, The Netherlands 30−31 October 2020 > ESPE Connect 2021 Copenhagen Denmark 6–8 May 2021 > ESPE Schools 2020 > Dates and deadlines We also thank John Gregory (UK), who has been Winter School Co-ordinator since 2014 and is stepping down this year. Justin Davies (UK) will be the new Co-ordinator for 2021
News
Resources
Patient leaflets Recently reviewed and updated patient information leaflets are now available on our website, covering diabetes, congenital adrenal hyperplasia, multiple pituitary hormone deficiency, craniopharyngioma, growth hormone deficiency, hypothyroidism and hyperthyroidism. Produced as part of an ESPE initiative led by Gary Butler (UK), they are also endorsed by the British Society for Paediatric Endocrinology and Diabetes and by the Child Growth Foundation. There are two readability levels: ‘Easy’ for patients and ‘Average’ for parents and guardians. Please use and share this resource (translations will be available in due course). www.eurospe.org/patients/ english-information-booklets ESPE e-Learning Supporting this issue’s theme: • Late Endocrine Effects of Treatment for Childhood Cancer − a chapter within Multiple Endocrine Deficits • Yu-Ting, a boy with leukemia (in Growth and Growth Regulation) − a case of small stature after oncological treatment. In addition: the Diabetes ISPAD Guidelines have been updated and many cases have been added. www.espe-elearning.org (registration is free)
Hot topics
Intro
Bringing you recent highlights from the world of research
Hot topics
Adiponectin receptor
agonist counteracts adverse
corticosterone effects
Systemic glucocorticoid (GC) treatment is the standard-of-care therapy for several autoimmune and inflammatory disorders. Because of its pleiotropic action, long term GC treatment can, however, lead to obesity, insulin resistance and osteoporosis, and induces growth retardation in children. Nicolas et al. used the adiponectin receptor agonist AdipoRon in young mice treated with corticosterone for 7 weeks, to test whether the negative effects of GC treatment could be ameliorated. Application of AdipoRon during the last 20 days of treatment reversed weight gain by acting mainly on intra-abdominal fat and on corticosterone-induced adipocyte hypertrophy. Increased leptin and insulin were normalised by AdipoRon, as well as corticosterone-induced hypoglycaemia. There was, however, no effect on lean mass, bone mass and, consequently, body length, which was decreased by corticosterone treatment. Since AdipoRon effectively reversed weight gain and partly normalised disturbances in glucose metabolism, it could be considered a potential therapeutic tool to counteract GC-induced side effects.
Hot topics
Obesity-associated pancreatic islet inflammation
Chronic, low grade inflammation is a characteristic of obesity and type 2 diabetes mellitus (T2DM). Ying et al. have reviewed the immune cell-mediated inflammation in pancreatic islets in these insulin-resistant states and the consequences thereof for β-cell function. In contrast to type 1 diabetes mellitus, the dominant immune cell type causing inflammation in obese and T2DM islets is the macrophage. The increased macrophage accumulation in T2DM islets primarily arises through local proliferation of resident macrophages, which then provide signals (such as platelet-derived growth factor) that drive β-cell hyperplasia (a classic feature of obesity). In addition, islet macrophages also impair the insulin secretory capacity of β-cells. Through these mechanisms, islet-resident macrophages underlie the inflammatory response in obesity and mechanistically participate in the β-cell hyperplasia and dysfunction that characterise this insulin-resistant state. These findings point to the possibility of therapeutics that target islet inflammation, to elicit beneficial effects on β-cell function and glycaemia
Hot topics
Teplizumab: a step towards
prevention of type 1 diabetes
mellitus
A phase 2, randomised, placebo-controlled, double-blind trial of teplizumab (an Fc receptor non-binding anti-CD3 monoclonal antibody) was conducted in relatives of patients with type 1 diabetes who were at high risk for development of clinical disease. The study included 76 participants (55 (72%) were <18 years of age), who were randomly assigned to receive a single 14-day course of either teplizumab (n=44) or placebo (n=32). Progression was assessed on the basis of oral glucose tolerance test at 6-monthly intervals. The disease was diagnosed in 19 (43%) of those who received teplizumab and in 23 (72%) of those who received placebo. The median time to the diagnosis was 48.4 months in the teplizumab group and 24.4 months in the placebo group. Thus, teplizumab delayed the progression of clinical disease in high risk participants. Further studies with a larger number of at risk subjects and frequent dosing are required to see if teplizumab will prevent type 1 diabetes mellitus completely.
Hot topics
Reduced mortality in carriers of CYP21A2 mutations
Congenital adrenal hyperplasia (CAH) due to 21-hydroxylase deficiency has an incidence of 1:10 000 to 1:20 000 in most populations. A survival advantage in carriers of CYP21A2 mutations is thought to contribute to the high incidence of CAH worldwide. A recent Swedish study investigated the mortality and cause of death in 1143 carriers of a CYP21A2 mutation (i.e. parents of CAH patients identified from the Swedish National CAH Registry). The results were compared with general population controls, matched for age and sex. The mortality and cause of death were investigated via the Swedish Cause of Death Registry. Carriers of a CYP21A2 mutation had overall reduced mortality and lower risk of death due to infection and, particularly, pneumonia. The lower mortality was more pronounced for carriers of severe mutations. Possible adaptive changes in the hypothalamic pituitary adrenal axis, with a more efficient cortisol response to stress in carriers of CYP21A2 mutations, could be a contributing factor to the reduced mortality
Feature
Feature
Late effects of haematopoietic
stem cell transplantation
Wassim Chemaitilly examines the endocrine complications of paediatric haematopoietic stem cell transplantation, and the value of systematic screening
Indications for paediatric haematopoietic stem cell transplantation (HSCT) include haematologic malignancies, non-malignant haematologic disorders and inborn errors of metabolism. Conditioning for HSCT requires high dose myeloablative chemotherapy with or without total body irradiation (TBI). Prolonged treatment with glucocorticoids may be necessary to treat graft versus host disease. Long term survivors may experience late-onset complications after a variable latency period. Endocrine late effects have been reported in 60% of survivors treated before the age of 10 years.1 Prevalence and risk factors Impaired linear growth has been reported in 20−80% of survivors. Individuals treated with TBI are at risk for growth hormone deficiency (GHD), especially if TBI was delivered at higher doses and/or in a single fraction. Younger age at HSCT and previous treatment with cranial radiotherapy are additional risk factors for GHD.2 Radiation-induced growth plate injury may further contribute to the risk of short stature.3 Among female survivors, 65−85% experience premature ovarian insufficiency, while 48−85% of males have azoospermia. This is due to the gonadotoxic nature of TBI and chemotherapy agents such as cyclophosphamide and busulfan. Males experience Leydig cell failure less frequently, unless they were previously treated with testicular irradiation.2 Up to 50% of survivors are diagnosed with primary hypothyroidism; TBI is the main risk factor. Transfer of autoimmune disease from the graft donor may further contribute to the risk.4 Survivors were reported to experience secondary thyroid cancer after a median latency time of 8.5 years post-TBI.5 Up to 21% of 5-year survivors have severe bone mineral density (BMD) deficit.6 Changes induced by leukaemia, exposure to TBI and/or glucocorticoids, endocrinopathies and lifestyle variables may contribute to the risk. Higher than expected rates of insulin resistance, impaired glucose tolerance and diabetes mellitus have been reported in survivors, especially following TBI. These complications frequently occur in the absence of obesity, and seem related to body composition alterations (decreased lean mass, increased body fat).7 Screening and management It is reasonable to initiate screening 1 year after HSCT in the absence of symptoms, using recommendations such as those provided by the Children’s Oncology Group.8 The management of endocrine disorders may follow guidelines used in non-cancer populations, with some adjustment to account for specific situations. Attention to body proportions and careful pubertal staging are of utmost importance. Skeletal irradiation disproportionately impacts spinal growth; this is best assessed by measuring and tracking sitting heights. Evaluation for GHD should include dynamic testing, as serum insulin-like growth factor-1 and insulin-like growth factor-binding protein-3 levels are not always reliable. GH replacement in survivors with GHD does not increase the risk of cancer recurrence or death from cancer; reported associations with secondary neoplasia are an area of active research.9 Treatment with GH may not allow full recovery of growth potential, given the frequency of other causes of poor growth.3 Treatment toxicity may impact testicular size without impairing testosterone secretion. Testicular volume is not a reliable indicator of puberty in male survivors. When in doubt, medical providers should measure morning serum testosterone levels.9 Fertility preservation via mature oocyte cryopreservation or sperm banking should be offered whenever feasible. Strategies using gonadal tissue cryopreservation are considered experimental, and may carry the risk of cancer cell re-seeding in individuals treated for haematologic malignancies.10 The use of ultrasound to screen for secondary thyroid cancer remains an area of controversy, given the lack of evidence supporting long term benefit when compared with periodic neck palpation by an experienced provider.11 There is a paucity of data to guide the frequency of BMD screening studies and the long term benefits of bisphosphonates.12 Similarly, it is unknown whether the use of glucose-lowering medications could delay the onset of diabetes mellitus in survivors with insulin resistance.7 In conclusion Endocrine late effects are common in survivors of paediatric HSCT. Systematic screening creates opportunities for early diagnosis and treatment. Interventional studies aiming at improving metabolic health are lacking in this population. Wassim Chemaitilly Department of Pediatric Medicine, Division of Endocrinology, St Jude Children’s Research Hospital, Memphis, TN, USA
Feature
Fertility preservation in
children and adolescents
with cancer
Rodrigo Romao considers the issues surrounding fertility
preservation for young people with cancer, and the
importance of raising the subject with patients and their
families.
A cancer diagnosis places an enormous burden upon paediatric patients and their families. Initial exchanges between families and the medical team will fittingly be devoted to treatment approaches, outcomes and the possibility of cure. Nonetheless, the overall positive survival outlook for paediatric oncologic patients compared with adults demands that long term treatment effects and survivorship issues be included in the discussion early on. The impact of cancer treatment on fertility is real and multifactorial. Surgical removal of gonads, chemotherapy (particularly with alkylating agents like cyclophosphamide) and radiation exposure can impact reproductive function and lead to infertility. Survivors of childhood cancer and their families want to know about these risks and what can be done to mitigate them. Some have argued that information about the potential for infertility and strategies for fertility preservation are part of the disclosure process of a new cancer diagnosis.1 Strategies for fertility preservation Fertility preservation strategies are variable, depending on age and sex. Sperm banking for post-pubertal males who are able to ejaculate is the most straightforward method, and should be considered in every male teenager diagnosed with cancer. In pre-pubertal boys, some centres have been offering a biopsy for testicular tissue cryopreservation, performed during the same anaesthetic as for other procedures, such as central line insertion, bone marrow biopsy and intra-thecal chemotherapy administration. While recent reports of viable pregnancies using cryopreserved pre-pubertal testicular tissue in rodents and primates are promising, this option remains experimental in humans at this stage.2 In post-pubertal females, embryo or oocyte cryopreservation are hindered by the need for ovarian hyperstimulation for at least 2−3 weeks, which can pose an often unacceptable delay in the start of cancer therapy. Experimental ovarian tissue cryopreservation is also possible in pre-pubertal girls; the need for intra-abdominal surgery, albeit minimally invasive (laparoscopy), represents an important drawback. Laparoscopic ovarian transposition is also an option, to move the gonads out of the pelvic radiation field.3 Barriers to fertility preservation Despite the increase in understanding about this topic, many barriers still prevent fertility preservation in children and adolescents with cancer being offered routinely. Lack of knowledge among treating physicians, coupled with a desire to start treatment as quickly as possible, can be addressed by education and awareness campaigns. Cost remains a major deterrent to fertility preservation, since upfront and maintenance fees for sperm and tissue banking are prohibitive to many families. Insurance coverage tends to be minimal to non-existent, from both private and public insurers. At the IWK Health Centre in Halifax (Nova Scotia, Canada), a multidisciplinary oncofertility committee has been created. It meets regularly and has developed educational materials to be shared with patients and treating physicians at the time of diagnosis. We also assist newly diagnosed patients with information and in navigating the process for banking specimens and/or tissues, when so desired. The field of fertility preservation for children and adolescents with cancer is still very much in its infancy, but it is unquestionable that significant advances have been made in the last few years, and great opportunities lie ahead. Rodrigo Romao Associate Professor of Surgery and Urology, IWK Health Centre, Dalhousie University, Halifax, Nova Scotia, Canada
Feature
Late effects of particle therapy
for CNS tumours
Eva Bezak, Myxuan Huynh, Mikaela Dell’Oro and
Thomas Merchant discuss the side effects of X-rays
versus proton therapy
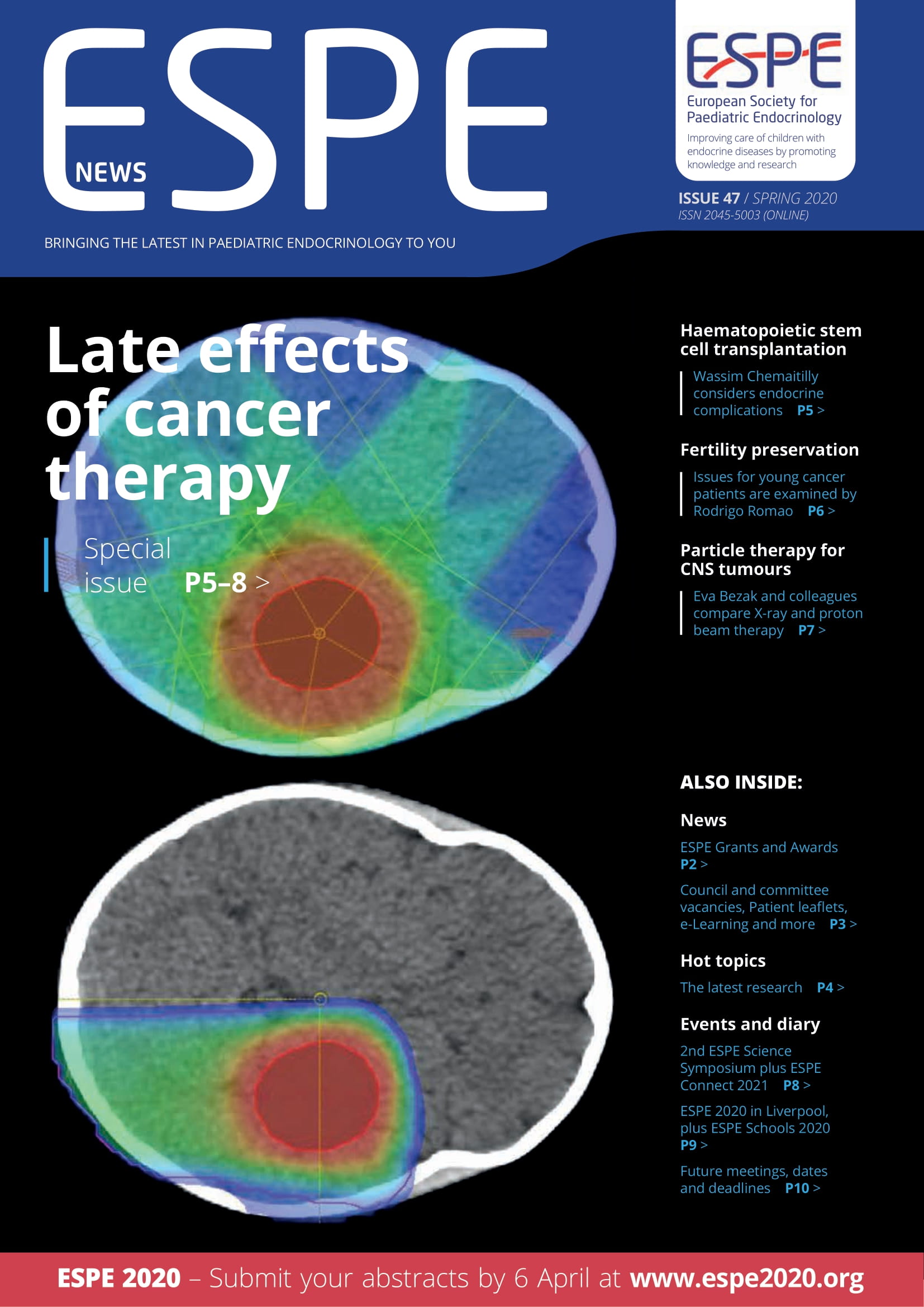
Paediatric tumours of the central nervous system (CNS) are the most common solid tumours, accounting for 20% of all cancers in children below the age of 15.1 In the USA alone, more than 500 new cases of medulloblastoma, the most common malignant brain tumour in children, are diagnosed annually. CNS tumours, and brain tumours more specifically, are the leading cause of cancer-related burden in childhood cancer survivors, due to their high incidence of mortality and long term effects which impact somatic, neurologic, endocrine and cognitive outcomes.2 Standard treatment strategies include a combination of surgery, chemotherapy and radiotherapy, all associated with complication risks. X-rays (photons) are the most commonly used radiation therapy, but protons and heavy ions (e.g. carbon ions) are of increasing interest, due to the superior properties of their dose distribution, resulting in more conformal dose delivery to the tumour, and much lower dose to healthy tissues compared with traditional X-ray irradiation (see Figure, and this issue’s cover). Questions remain regarding the long term effects of proton radiation therapy on paediatric patients. X-ray radiotherapy Paediatric brain tumour patients who undergo radiotherapy using photons experience a wide range of short and long term side effects. Short term side effects include nausea, vomiting, loss of appetite, headache, fatigue and hair loss.2 Paediatric CNS cancer patients are also at increased risk of hormonal and growth problems, cognitive development deficits, and probability of developing second malignancies later in life.2 Up to 62% of childhood cancer survivors report one or more radiation related late toxicities, with 33% being severe.3 Hearing loss impacts patient quality of life (QoL), communication skills, and social development,4 occuring with a latency of approximately 4 years. It is typically considered to be radiation-induced, though radiation is known to potentiate early ototoxicity effects of cisplatin.4 Neurocognitive effects of radiation therapy are associated with reductions in academic performance, employment rates and education opportunities in tumour survivors.2 Survivors often report decreased QoL due to difficulties in attention and cognitive processing speed.2 Cranial irradiation is associated with an IQ decline of 2−4 points per year.5 Risk factors for increased neurocognitive decline include younger age during treatment (<3 years old), large irradiated volumes and higher dose delivered.5 Deficits are known to correlate with the volume of cerebral white matter irradiated, particularly in the supratentorial brain, temporal lobe and hippocampal dentate gyrus.3 Up to 43% of survivors reported endocrine deficits, including osteoporosis, hypothyroidism and growth hormone deficiency (GHD).3 Total doses ≤30Gy to the pituitary have been associated with GHD in approximately 30% of patients, whilst mean doses ≤16.1Gy to the hypothalamus resulted in GHD in 50% of patients.3 Reductions in gonadotrophins and adrenocorticotrophic and thyroid-stimulating hormones have also been reported.3 These endocrine deficits correlate with high total doses to the hypothalamic-pituitary axis, and are increased when patients are treated at a younger age.3 All paediatric malignancies are associated with increased risks of second cancers following radiation therapy.6 X-ray therapy is associated with the development of solid malignancies, such as thyroid neoplasms and sarcomas, particularly in the treatment of CNS tumours such as gliomas and meningiomas.3 Proton therapy Proton therapy has been found to provide survival and tumour control outcomes for paediatric CNS cancers comparable with photon therapy, with reduced incidence of severe acute and late toxicities. This includes reduced severity of endocrine and neurological deficits. Papers comparing proton and photon cohorts also reported reduced haematological and gastrointestinal toxicities,7 improved QoL scores,8 and reduced IQ changes with the use of proton therapy.5 Reduced incidence of second cancers with proton therapy has also been reported.6 While the technology is still fairly novel and data collection is still in progress, the literature reports no significant reduction in IQ scores from baseline to follow up with the use of proton therapy.9−13 However, increased risks of neurological decline have been associated with patients <7 years of age, high doses to the left temporal lobe or hippocampus, infratentorial tumour location, medulloblastoma/pancreatic neuroendocrine tumour histology, larger tumour size, and longer time since radiation therapy.5,9 A significant decline in processing speed scores by a mean of 5.2 points (despite no significant change in overall full scale intelligence quotient (FSIQ) scores) was also reported.12 Another study reported an overall decrease in FSIQ of 1.5 points per year, related to decreases in processing speed and verbal comprehension index with the use of proton therapy.14 Reported endocrine deficits as a result of proton therapy ranged from grade 0 to grade 2. The majority of reported endocrine deficits included GHD or hypothyroidism,10,11,13-18 while others included adrenal/ cortisol deficiency and sex hormone deficiency.10,14 Doses >40Gy to the hypothalamus and pituitary correlated with the incidence of neuroendocrine deficits.9,14 73% of patients receiving ≥40Gy to the hypothalamus reported neuroendocrine deficits, compared with 44% in patients receiving doses <40Gy.14 While evidence exists in support of proton therapy for paediatric CNS patients, it is still scarce. Data are often difficult to analyse, as some studies used mixed patient cohorts (adults and children). Frequently, no distinction is made between higher risk patients (<3 years of age) and the main cohort. There are large variations in patients’ overall treatments (e.g. surgery extent, details of chemotherapy regimens), which all have compounding or even synergistic effects on toxicities. Results are also affected by insufficient durations of follow up, thus limiting the validity and reliability of reported literature. However, the reports are positive and promising, and the medical specialists involved in the treatment of childhood brain tumour patients are eager to learn of high quality evidence for the use of proton therapy across all tumour types. Eva Bezak, Myxuan Huynh and Mikaela Dell’Oro Cancer Research Institute and School of Health Sciences, University of South Australia and Royal Adelaide Hospital, Adelaide, SA, Australia Thomas Merchant St Jude Children’s Research Hospital, Memphis, TN, US
Events
Events
ESPE Science Symposium
CAH: from molecular medical research to clinical application 30–31 October 2020 Nijmegen, The Netherlands Register now for ESPE’s second Science Symposium, featuring interactive lectures by renowned scientists from around the world, along with discussion and networking time. Clinicians and researchers will discover the very latest developments in congenital adrenal hyperplasia (CAH), and make new connections. • €80 including dinner on the first night • 25 free registrations available to young clinicians and scientists-in-training • Hosted by Hedi L Claahsen and her team at the Radboud DSD Centre • Supported by the Endo-ERN and the ESPE DSD Working Group Apply by 1 June 2020 Programme and registration: www.espe-science-symposium-2020.com
Events
Lifelong endocrine care through
collaboration, discovery and innovation
10−12 September 2020, Liverpool, UK Many conditions we treat will affect our patients for the rest of their lives, and the 59th ESPE Meeting will therefore focus on lifelong care. We will also examine exciting discoveries in genetic/genomic research, leading to innovative approaches to treatment. Most importantly, the Meeting will be about friendships, networking and collaboration – both new and existing. The ACC, in the vibrant and beautiful Albert Dock area of Liverpool, is the venue for our Meeting: just 15 minutes’ walk from the city centre. Liverpool is one of the great cities of the world, with the attractions and infrastructure to cater for all, whilst also brimming with culture and heritage. Nearby Alder Hey Children’s Hospital is one of Europe’s biggest and busiest children’s hospitals, widely recognised as a world leader in children’s healthcare and research. We warmly welcome you to participate in a Meeting that will be rich in basic and clinical science, and will allow delegates from around the world to present and discuss their latest findings in an interactive and convivial environment Prizes and grants Submit your abstract to ESPE 2020 for the chance to win one of the following awards: • Henning Andersen Prizes • ESPE President Poster Awards • Travel Grants www.eurospe.org/grants awards/espe-meeting grants Fraudulent registration websites Please be aware that fraudulent websites have been in operation, selling fake registration to ESPE 2020. The ESPE website (at www.eurospe. org or www.espe2020.org) is the only official website where you can register to attend the ESPE Meeting.
[contact-form-7 id="520" title="Comments"]
